Morphological characteristics of the myocardium against the background of the altered content of the immune cells in the brain in mice of different strains with experimental parkinsonism
Main Article Content
Abstract
The aim – to investigate the features of structural changes in the myocardium in mice of different H-2 genotypes (human HLA analogue) with an experimental toxic model of parkinsonism against the background of changes in the content of lymphocytes and macrophages in the brain.
Materials and methods. Adult (6-7 months of age) male mice of the FVB/N (genotype H-2q) and 129/Sv (genotype H-2b) strains were injected once with the neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) at a dose of 30 mg/kg (research groups) or 0.9 % sodium chloride solution (control groups). In 18 days after injections, histological examination of the myocardium, immunophenotyping of brain cells for markers CD3 and CD11b were performed in mice of all experimental groups as well as the activity of intracellular myeloperoxidase (MPO) as a marker of the brain inflammatory process was assessed.
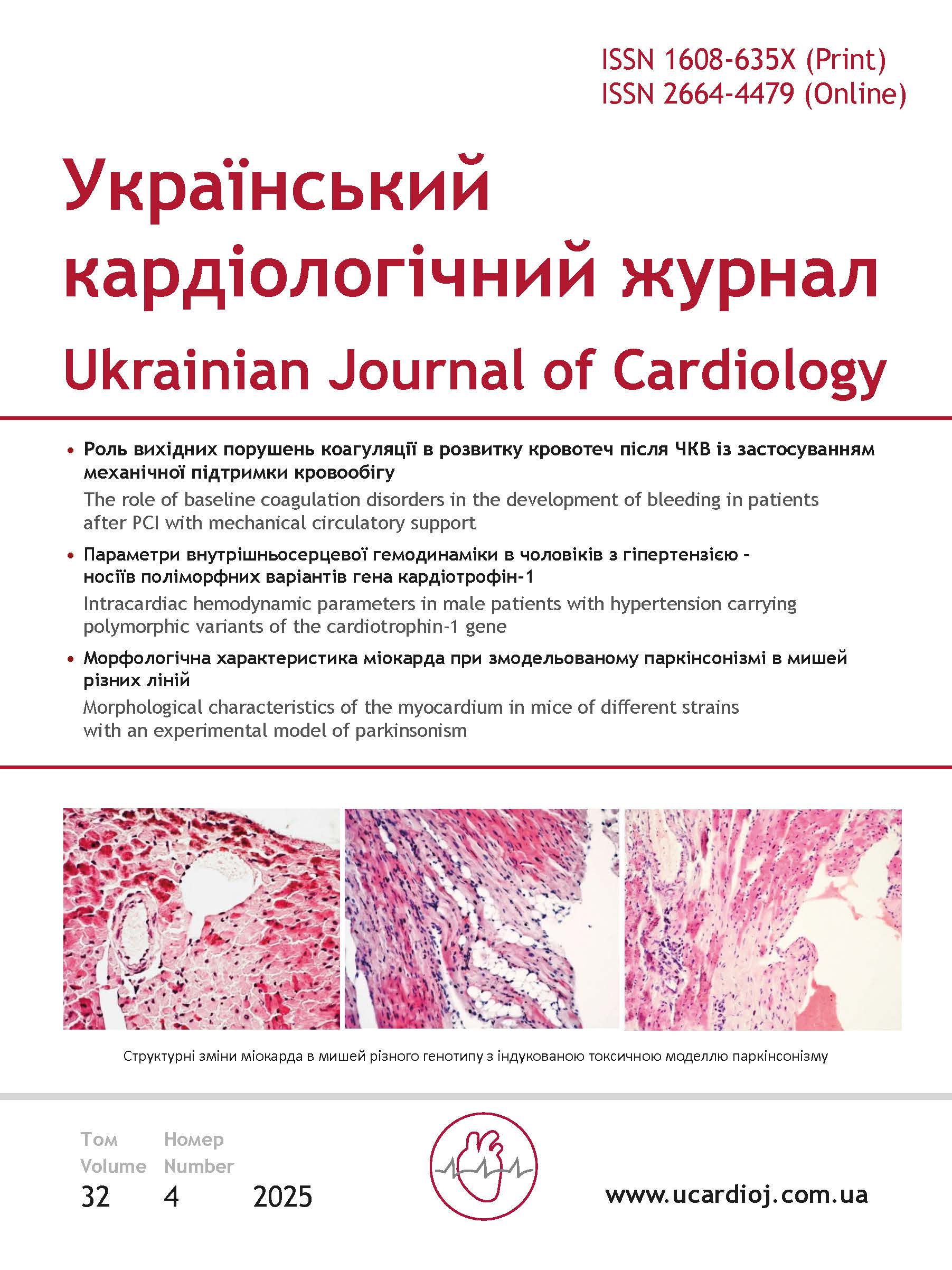
Results and discussion. After the administration of MPTP in the myocardium of the 129/Sv mice strain we have observed death of the cardiomyocytes with the appearance of adipose tissue and areas of sclerosis, the development of inflammation with signs of lymphocytic and macrophage infiltration as well as activation of fibroblasts and destructive changes in the blood vessel wall with the destruction of the endothelial cell monolayer. After MPTP administration in the myocardium of FVB/N mice strain we have observed cardiomyocyte death, accumulation of lymphocytes and fibroblasts and areas of fat embolism in the blood vessels. In mice of the 129/Sv strain, which were MPTP injected, the content of CD3+CD11b+ cells has been higher than in the control group mice. In research group of the FVB/N mice, the content of not only CD3+CD11b+ cells, but also CD3+ cells has exceeded the values of the indicators in the control animal group. After the injection of MPTP, the activity of MPO in the brain of the 129/Sv mice has been significantly higher than in control animals, but did not change in mice of the FVB/N strain.
Conclusions. In mice with a toxic model of parkinsonism, we have observed the myocardial damage, the manifestations and severity of which largely depended on the H-2 genotype of animals and peculiarities of accumulation of the immune system cells in this organ. We have established the differences in the balance of neuro-inflammatory marker cells (T lymphocytes, macrophages) as well as in the activity of the inflammatory marker MPO in the brain of research mice with different genotypes H-2. The experimental toxic model of parkinsonism may be useful for studying the development of combined myocardial pathology in parkinsonism involving immune system cells.
Article Details
Keywords:
References
Karaban IN, Shalenko OV, Kryzhanovskiy SA. Non-motor symptoms in clinical picture of the Parkinson’s disease. International neurological journal. 2017 Jan;1:58-63. https://doi.org/10.22141/2224-0713.1.87.2017.96538
Simon DK, Tanner СM, Brundin P. Parkinson disease epidemiology, pathology, genetics and pathophysiology. Clin Geriatr. 2020 Feb;36(1):1-12. https://doi.org/10.1016/j.cger.2019.08.002
Grosu L, Grosu AI, Crisan D, Zlibut A, Perju-Dumbrava L. Parkinson’s disease and cardiovascular involvement: Edifying insights. Biomedical reports. 2023 Feb;18(25):1-8. https://doi.org/10.3892/br.2023.1607
Potashkin J, Huang X, Becker C, Chen H, Foltynie Th, Marras C. Understanding the links between cardiovascular disease and Parkinson’s disease. Mov Disord. 2020 Jan;35(1):55–74. https://doi.org/10.1002/mds.27836
Cunca-Bermejo L, Almela A, Navarro-Zaragora J, Ferbabdez Villalba E, González-Cuello AM, Laorden ML, Herrero MT. Cardiac changes in Parkinson’s disease: Lessons from clinical and experimental evidence. Int J Mol Sci. 2021 Dec;22:13488. https://doi.org/10.3390/ijms222413488
Stephenson E, Savvatis K, Mohiddin SA, Marelli-Berg M. T-cell immunity in myocardial inflammation: pathogenic role and therapeutic manipulation. British Journal of Pharmacology. 2017 Sep;174:3914–3925. https://doi.org/10.1111/bph.13613
Chen R, Zhang H, Tang B, Luo Y, Yang Y, Zhong X, Chen S, Xu X, Huang Sh, Liu C. Macrophages in cardiovascular diseases: molecular mechanisms and therapeutic targets. Signal Transduction and Targeted Therapy. 2024 May;9:130. https://doi.org/10.1038/s41392-024-01840-1
Dalton KR, Kidd CJ, Hack N. Toxin Induced Parkinsonism and Hospitalization Related Adverse Outcome Mitigation for Parkinson’s Disease: A Comprehensive Review. J. Clin. Med. 2023 Jan;12:1074. https://doi.org/10.3390/jcm12031074
Albakri A. Toxin-induced cardiomyopathy: A review and pooled analysis of pathophysiology, diagnosis and clinical management. Res Rev Insights. 2019 Feb;3:1-21. https://doi.org/10.15761/RRI.1000150
Putiy YV, Fuchko OL, Vaschenko NM, Rozova KV. Formation of ultrastructural adaptive reactions in body tissues under experimental parkinsonism. Physiological J. 2023 Dec;69(3):74-82. https://doi.org/10.15407/fz69.03.074. Ukrainian.
Liu X, Wei B, Bi Q, Sun Q, Li L, He J, Weng Y, Zhang S, Mao G, Bao Y, Wan S, Shen X, Yan J, Shi P. MPTP-induced impairment of cardiovascular function. Neurotox Res. 2020 Jun; 38:27-37. https://doi.org/10.1007/s12640-020-00182-4
Labunets IF, Panteleymonova TM, Utko NO, Kyryk VM, Savosko SI, Litoshenko ZL. Changes in the number of macrophages, T-lymphocytes, activity of antioxidant enzymes in the brain, behavior and structure of the central nervous system neurons in adult and aging mice of different strains with the MPTP-induced model of parkinsonism. International Neurological J. 2023 Dec;19(4):119-128. https://doi.org/10.22141/2224-0713.19.4.2023.1010. Ukrainian.
Kannarkat GT, Cook DA, Lee JK, Chang J, Chung J, Sandy E, Paul KC, Ritz B, Bronstein J, Factor SA, Boss JM, Tansey MG. Common genetic variant association with altered HLA expression synergy with pyrethroid exposure and risk for Parkinson’s disease: an observational and case control study. Parkinson’s disease. 2015 Apr;1:15002. https://doi.org/10.1038/npjparkd.2015.2
Lomakovsky OM, Minchenko ZM, Havrylenko TI, Pidhajna OA. Immunogenetic features of the HLA system in patients with early and late development of coronary heart disease and previous myocardial infarction. J Rheumatology. 2021 Dec;86(4):80-87. https://doi.org/10.32471/rheumatology.2707-6970.86.16547 Ukrainian.
Labunets IF, Rodnichenko A. The state of the immune and endocrine systems in mice with different H-2 haplotypes and its potential relationship with the manifestations of experimental parkinsonism. Physiological J. 2024 Nov;70(3):42-50. https://doi.org/10.15407/fz70.03.042. Ukrainian.
Labunets IF, Utko NA, Savosko S, Panteleymonova TN, Butenko GM. Changes in nigral neuronal structure, indices of antioxidant protection of the brain and behavior in mice of different age with MPTP parkinsonism model. International Neurological J. 2020 Nov;16(3):7-15. https://doi.org/10.22141/2224-0713.16.3.2020.203444. Ukrainian.
Zeng XS, Geng WS, Jia JJ. Neurotoxin-induced animal models of Parkinson disease: pathogenic mechanism and assessment. ASN Neuro. 2018 Jan;10(1). https://doi.org/10.1177/1759091418777438.
Kyryk V, Ustymenko A, Klymenko P, Tsupykov O. Development and Validation of a Lipopolysaccharide-Induced Myocardial Inflammation Model in Mice for Preclinical Research on Stem Cell-based Therapy. Anti Aging East Eur. 2024 Nov;3(4):221-232. https://doi.org/10.56543/aaeeu.2024.3.4.09. Ukrainian.
Prysiazhniuk AI, Rudyk MP, Chervinska TM, Dovbynchuk TV, Opeida IV, Skivka LM, Tolstanova GM. Role of peripheral dopaminergic system in the pathogenesis of experimental colitis in rats. Ukr Biochem. 2017 Oct;89(4):56-67. https://doi.org/10.15407/ubj89.04.056. Ukrainian.
Pulli B, Ali M, Forghani R, Schob S, Hsieh KL, Wojtkiewicz G, Linnoila JJ, Chen JW. Measuring myeloperoxidase activity in biological samples. PLoS One. 2013 Jul 5;8(7):e67976. https://doi.org/10.1371/journal.pone.0067976
Jones GLA, Sang E, Golddard C, Mortishire-Smith C, Sweatman BC, Haselden JN, Davies K, Grace AA, Clarke K, Griffin J-L. A functional analysis of mouse model of cardiac disease through metabolic profiling. JBC. 2005;280(9):7530-7539. https://doi.org/10.1074/jbc.M410200200
Boykiv AB. Changes in humoral immunity in animals with adrenal myocardiopathy during different types of inflammatory reactions. Bulletin of Scientific Res. 2008 Nov;3:60-63. Ukrainian.
Kovalenko VM, Nesukay EG, Talaieva TV, Sychov OS, Cherniuk SV, Kirichenko RM, Tretiak IV. Features of the debut of acute myocarditis in patients after COVID-19 infection. World Med and Biology. 2023 Aug;2(84):87-92. https://doi.org/10.26724/2079-8334-2023-2-84-87-92 Ukrainian.
Prevete N, Sorriento D. The Role of Macrophages in Cardiac Function and Disease. J Mol Pathol. 2023 Oct;4:318-332. https://doi.org/10.3390/jmp4040026
Labunets I, Rodnichenko A, Savosko S, Pivneva T. Reaction of different cell types of the brain on neurotoxin cuprizone and hormone melatonin treatment in young and aging mice. Front Cell Neurosci. 2023 Apr;17:1131130. https://doi.org/10.3389/fncel.2023.1131130
Appel H, Beers R, Henkel S. The T cell-microglial dialogue in Parkinson’s disease and amyotrophic lateral sclerosis: are we listening? Trends Immunol. 2010 Jan;31(1):7-17. https://doi.org/10.1016/j.it.2009.09.003
Frosch M, Amann L, Prinz M. CNS-associated macrophages shape the inflammatory response in a mouse model of Parkinson’s disease. Nat Commun. 2023 Jun;14(1):3753. https://doi.org/10.1038/s41467-023-39061-9
Kim HAh, Whittle SC, Lee S, Chu HX, Zhang ShR, Wei Z, Arumugam TV, Vinh A, Drummond GR, Sobey CHG. Brain immune cell composition and functional outcome after cerebral ischemia: comparison of two mouse strains. Front Cell Neurosci. 2014;8. Article 365 https://doi.org/10.3389/fncel.2014.00365